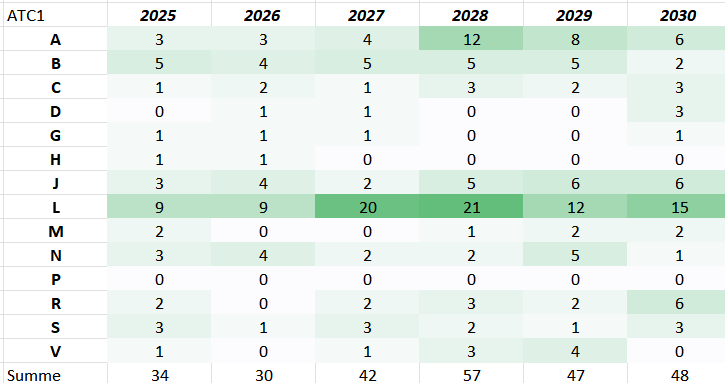
Patent Expiries 2025-2030
Expected generic entries by indication area in the next years on the pharmaceutical market Germany
Generic entry after expiration patent / SPC / data protection
For several ATC codes, only 1 entry was counted per active ingredient (combination), only the first expiry for several SPCs
| Code | Contents |
|---|---|
| A | Alimentary tract and metabolism |
| B | Blood and blood forming organs |
| C | Cardiovascular system |
| D | Dermatologicals |
| G | Genito-urinary system and sex hormones |
| H | Systemic hormonal preparations, excluding sex hormones and insulins |
| J | Antiinfectives for systemic use |
| L | Antineoplastic and immunomodulating agents |
| M | Musculo-skeletal system |
| N | Nervous system |
| P | Antiparasitic products, insecticides and repellents |
| R | Respiratory system |
| S | Sensory organs |
| V | Various |
(List from: "ATC: Structure and principles". WHO Collaborating Centre for Drug Statistics Methodology)
Over the next 6 years, generics products for more than 200 active ingredients / drug combinations can be expected, including products like Eylea, Eliquis, Forxiga, Trulicity, IMBRUVICA, OPDIVO. Ocrevus, Entresto,…


